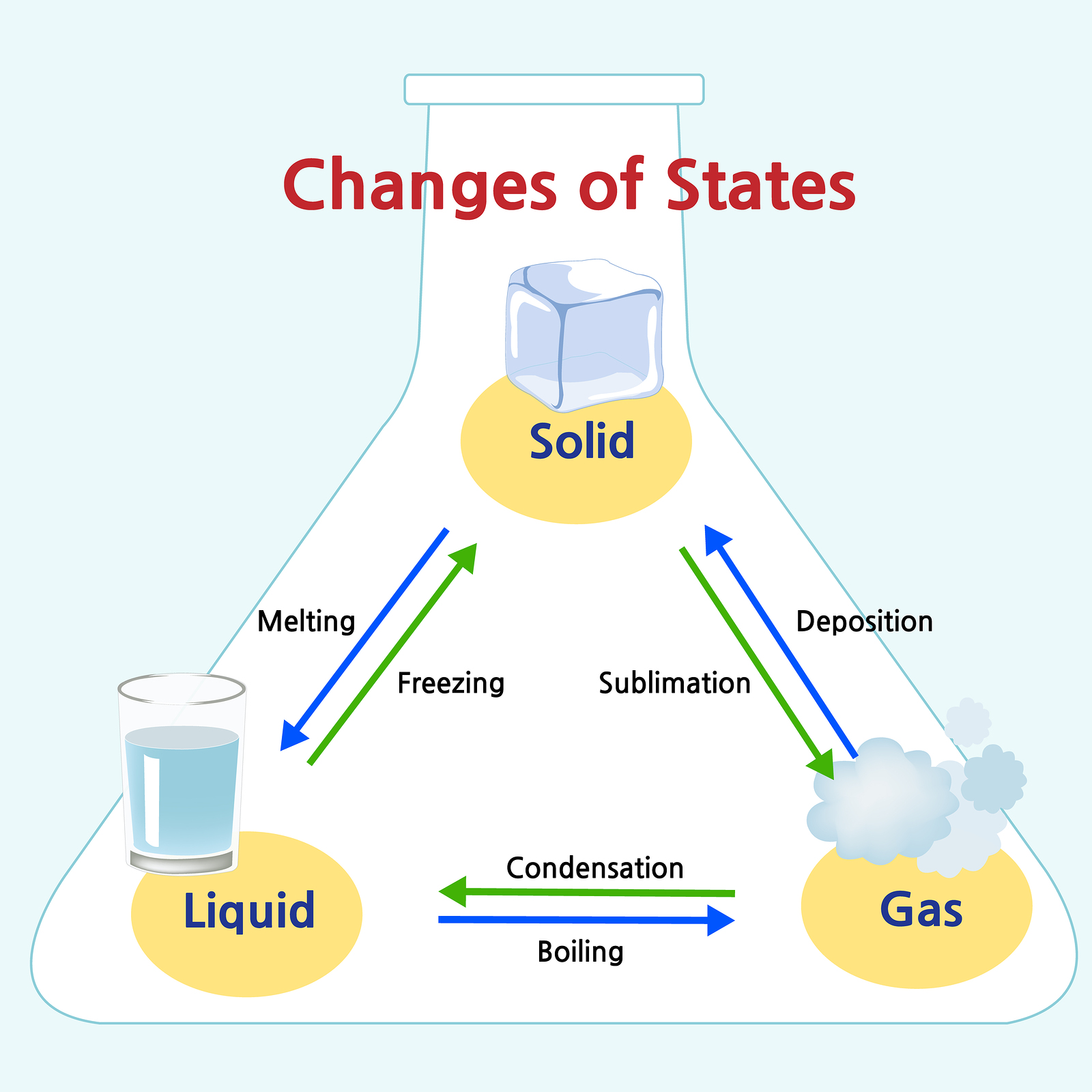
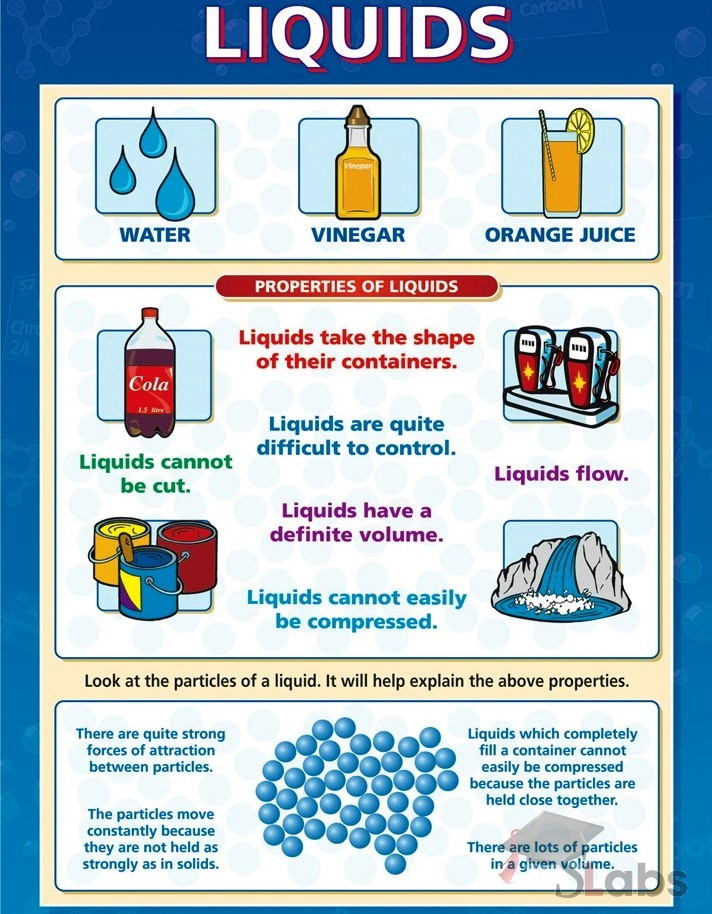
Solid Liquid Gas Chart
Solid Liquid Gas Chart - In this state, the distinction between liquid and gas disappears. For example, the addition of heat can melt ice into liquid water and turn that water into. Solids are incompressible and have high density, compared to liquids and gases. The particles vibrate back and forth within their fixed positions and do not move freely. Web a vapor can exist in equilibrium with a liquid (or solid), in which case the gas pressure equals the vapor pressure of the liquid (or solid). They vibrate and move freely at high speeds. Like a liquid, a gas takes the shape of a container. Where solids and liquids differ is in their shape. Density very low density of a substance in usually lower in the liquid than in the solid state. Gas refers to a state of matter, do not have any shape but conform to the shape of the container, completely, in which it is put in. Web they don’t pour like a liquid. Web aligned with the topic properties of the three states of matter, the chart here stimulates interest, summarizes the properties of solids, liquids and gases and assists in distinguishing between them. A gas will fill any container, but if the container is not sealed, the gas will escape. Relate the interaction potential to the forces. In ancient greece, one philosopher recognized how water could change form and reasoned that everything must be made of water. Liquid vibrate, move about, and slide past each other. Gas refers to a state of matter, do not have any shape but conform to the shape of the container, completely, in which it is put in. The state that a given substance exhibits is also a physical property. Often the state of matter of a substance may be changed by adding or removing heat energy from it. Matter is usually classified into three classical states. Web aligned with the topic properties of the three states of matter, the chart here stimulates interest, summarizes the properties of solids, liquids and gases and assists in distinguishing between them. Matter is usually classified into three classical states. Highly strong intermolecular forces between the molecules, leads to a definite volume in solids. Web reimagine the everyday with a closer. In the video here, sal uses a horizontal line through the phase diagram. The state that a given substance exhibits is also a physical property. Web they don’t pour like a liquid. Relate the interaction potential to the forces. Quartz (solid), water (liquid), nitrogen dioxide (gas). A gas is a state of matter lacking either a defined volume or defined shape. They can be crystalline, like table salt, or amorphous, like glass, rubber or plastic. Solid (the ice), liquid (the water) and gas (the vapor) are the three most common states of matter — at least on earth. Web this is another great question. Web gas. Matter occurs in four states: A supercritical fluid (scf) is a gas whose temperature and pressure are above the critical temperature and critical pressure respectively. Gas vibrate and move freely at high speeds. In ancient greece, one philosopher recognized how water could change form and reasoned that everything must be made of water. Where solids and liquids differ is in. A supercritical fluid (scf) is a gas whose temperature and pressure are above the critical temperature and critical pressure respectively. Matter occurs in four states: Density very low density of a substance in usually lower in the liquid than in the solid state. Web a vapor can exist in equilibrium with a liquid (or solid), in which case the gas. Thus, there is no definite volume. For example, the addition of heat can melt ice into liquid water and turn that water into. Liquid vibrate, move about, and slide past each other. Web aligned with the topic properties of the three states of matter, the chart here stimulates interest, summarizes the properties of solids, liquids and gases and assists in. Liquid vibrate, move about, and slide past each other. Quartz (solid), water (liquid), nitrogen dioxide (gas). Solid (the ice), liquid (the water) and gas (the vapor) are the three most common states of matter — at least on earth. Web examples of liquids include water, juice, and vegetable oil. Relate the interaction potential to the forces. A supercritical fluid (scf) is a gas whose temperature and pressure are above the critical temperature and critical pressure respectively. Liquid vibrate, move about, and slide past each other. No expand when heated expand greatly when heated. Where solids and liquids differ is in their shape. For example, the addition of heat can melt ice into liquid water and turn. Direct the children of grade 2 and grade 3 to observe the illustrations given in this circle. However, water isn’t the only type of matter that. Web examples of liquids include water, juice, and vegetable oil. Web gas are well separated with no regular arrangement. But, it doesn't have to be horizontal. Web matter typically exists in one of three states: Liquid vibrate, move about, and slide past each other. Web difference between solid liquid and gases; Highly strong intermolecular forces between the molecules, leads to a definite volume in solids. Often the state of matter of a substance may be changed by adding or removing heat energy from it. Liquid is a substance, that flows freely, having a definite volume but no permanent shape. Web this is another great question. Add or remove heat and watch the phase change. They can be crystalline, like table salt, or amorphous, like glass, rubber or plastic. Where solids and liquids differ is in their shape. Gas refers to a state of matter, do not have any shape but conform to the shape of the container, completely, in which it is put in. Direct the children of grade 2 and grade 3 to observe the illustrations given in this circle. A gas will fill any container, but if the container is not sealed, the gas will escape. Web matter typically exists in one of three states: Web compare three states of matter: Web solids and liquids have a fair bit in common, as in both states the molecules are joined together. No expand when heated expand greatly when heated. But, it doesn't have to be horizontal. Solids, liquids, gases, and plasma. As long as you are at 100 c, you can change the phase by changing the pressure on the system. Expand a little when heated.Solids, Liquids, Gases Wall Chart Rapid Online
States of Matter NurseHub
What are states of matter? TheSchoolRun
Solids, Liquids, Gases Chart Scholars Labs
Solids, Liquids, Gases Chart Scholars Labs Chemistry classroom
Solids, Liquids, & Gases! Rachel A Tall Drink of Water
States of Matter
Solids Liquids And Gases Poster
States of Matter Anchor Chart Classroom Decor Posters for Solid Liquid
Learning about matter and its various states. Anchor charts, Solid
A Supercritical Fluid (Scf) Is A Gas Whose Temperature And Pressure Are Above The Critical Temperature And Critical Pressure Respectively.
Gas Can Be Compressed Much More Easily Than A Liquid Or Solid.
Density Very Low Density Of A Substance In Usually Lower In The Liquid Than In The Solid State.
A Gas Is A State Of Matter Lacking Either A Defined Volume Or Defined Shape.
Related Post: